
New Vaccine, Promising Pathway to Malaria Elimination on Bioko Island, EG
The following news release was issued by Sanaria on April 10, 2019, and reprinted with their permission.
For more information, read the article "Promising malaria vaccine to be tested if first large field trial" published in Nature International Journal of Science.
Please note our forward-looking statements disclaimer.
Sanaria Inc. and the Government of Equatorial Guinea signed an agreement that continues their joint commitment to develop Sanaria® PfSPZ Vaccine as a tool for malaria elimination. This follows a new, broader agreement between the Government of Equatorial Guinea (EG) and industry partners, Marathon Oil, Noble Energy Inc. and Atlantic Methanol Production Company (AMPCO), to sponsor clinical development of Sanaria® PfSPZ Vaccine, including the Phase 3 clinical program starting in 2020.
This long standing, successful partnership highlights the unique involvement of an African government and private-sector energy companies in developing a vaccine for a largely African problem. The larger funding program, the Bioko Island Malaria Elimination Program (BIMEP), is targeting elimination of malaria from a population of over 280,000 on Bioko Island by integrating PfSPZ Vaccine into standard anti-malaria activities, including mosquito control and malaria diagnosis and treatment, coordinated by Medical Care Development International.
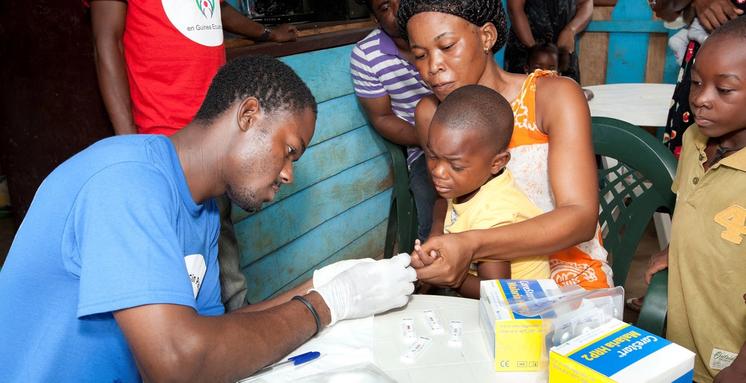
Malaria is a devastating disease of almost unfathomable impact, which according to the World Health Organization caused an estimated 219 million clinical cases and 435,000 deaths in 2017. Malaria directly reduces the Gross Domestic Product of African countries by at least 1% annually.
“Our goal is licensure and deployment of a vaccine to prevent malaria. We are excited to take our next step with the Government of Equatorial Guinea, Marathon Oil, Noble Energy and AMPCO on the pathway to PfSPZ Vaccine licensure and then malaria elimination on Bioko Island. This unique partnership continues to show the EG Government’s commitment to being the first African government to invest significant funds in development of a malaria vaccine,” said Stephen L. Hoffman, CEO of Sanaria.
The Republic of Equatorial Guinea, through the Ministries of Mines and Hydrocarbons, and Health and Social Welfare, and its oil and gas sector partners will provide $22 million USD to support vaccine development and malaria control efforts within BIMEP for 2019, and the Government of EG, the major financial contributor in this initiative, will further provide $4.5 million over the next three years to directly support Sanaria’s vaccine development efforts. These funds are in addition to the already-expended $48 million for the Equatoguinean Malaria Vaccine Initiative.
Following three previous clinical trials, the next step is a Phase 3 clinical trial in Malabo, EG, which will provide data to support submission to US and European regulatory agencies for licensure of PfSPZ Vaccine. BIMEP is also supported by the Ifakara Health Institute (IHI) (Tanzania) and the Swiss Tropical and Public Health Institute.
Dr. Salim Abdulla, former IHI Director, recognized that “although other African governments have strong malaria control programs, none have made the financial commitment to malaria vaccine development made by the EG Government. This program has also supported the human and infrastructure capacity building critical for the Phase 3 clinical and malaria elimination programs.”
The Vice Minister of Health and Social Welfare, Mitoha Ondo’o Ayekaba further stressed that, “this program also makes important contributions toward modernizing the healthcare system in the country, providing much needed training and state-of-the-art facilities on top of a unique vaccine development program.”

Documentary Film Hope for Bioko Watch the video
About Sanaria Inc.:
Sanaria is a socially focused, for-profit company. Sanaria’s mission is to develop and commercialize whole-parasite vaccines that confer protection against malaria.
About BIMEP:
The Bioko Island Malaria Elimination Program (BIMEP) is a merger of the Bioko Island Malaria Control Program and the Equatoguinean Malaria Vaccine Initiative, that in parallel have reduced malaria prevalence from 45% to 12.5% in 15 years, and established clinical research capacity in Equatorial Guinea (EG) by developing personnel, infrastructure, regulatory processes, ethical review, a clinical laboratory and a national research institute. BIMEP plans to eliminate malaria from Bioko Island (population of >280,000) using PfSPZ vaccines, before moving efforts to mainland EG and other parts of Africa.
Forward-Looking Statements
This press release contains forward-looking statements. All statements that address operating performance, events or developments that we expect or anticipate will occur in the future are forward-looking statements. These forward-looking statements are based on management's beliefs and assumptions and on information currently available. You should not place undue reliance on any such forward-looking statements because such statements speak only as of the date when made. We do not undertake any obligation to publicly update or revise any forward-looking statements, for any reason, except as required by law. Forward-looking statements are subject to certain risks and uncertainties that could cause actual results, events and developments to differ materially from our historical experience and our present expectations or projections.























